

Gilead Sciences Statement on Request to Rescind Remdesivir orphan Drug Designation Gilead has submitted a request to the U.S. Food and Drug Administration to rescind the orphan drug designation it was granted for the investigational antiviral remdesivir for the treatment of COVID-19 and is waiving all benefits that accompany the designation. Gilead is confident that it can maintain an expedited timeline in seeking regulatory review of remdesivir, without the orphan drug designation. Recent engagement with regulatory agencies has demonstrated that submissions and review relating to remdesivir for the treatment of COVID-19 are being expedited.
In early March, Gilead sought and was subsequently granted an orphan drug designation for the remdesivir as a potential treatment for COVID-19. orphan drug designation is granted by the FDA in situations where the disease affects fewer than 200,000 patients in the United States.
Among the benefits of orphan drug designation, this status results in a waiver of the requirement to provide a pediatric study plan prior to the submission of a New Drug Application â a process that can to take up to 210 days to review.
Gilead recognizes the urgent public health needs posed by the COVID-19 pandemic. The company is working to advance the development of remdesivir as quickly as possible, and will provide updates as they become available.
æ¥åï¼åå©å¾·ç§å¦å
¬å¸çå¨ç æç
æ¯çæ³ç德西é¦ï¼remdesivirï¼è·å¾ç¾å½FDAæäºçå¤å¿è¯èµæ ¼ï¼orphan drug designationï¼ï¼éåºç为æ°å ç
æ¯ç
ï¼COVID-19ï¼ãè¿ä¸æ¶æ¯å¼èµ·äºä¸å°äºè®®ï¼æäºä¸ç人士æ
å¿å¤å¿è¯èµæ ¼å¯è½ä¼å½±åç德西é¦çå¯åæ§ãä»æ¥ï¼åå©å¾·ç§å¦ï¼Gilead Sciencesï¼å
¬å¸åå¸å£°æï¼å®£å¸å·²ç»åç¾å½FDAæåºç³è¯·ï¼è¦æ± FDAæ¶åæäºç德西é¦çå¤å¿è¯èµæ ¼ï¼å¹¶ä¸æ¾å¼ä¸å¤å¿è¯èµæ ¼ç¸å
³çææä¼æ æçã
åå©å¾·ç§å¦è¡¨ç¤ºï¼æä¿¡å¿å³ä½¿å¨æ²¡æå¤å¿è¯èµæ ¼çæ åµä¸ï¼ä»è½ä¿æç德西é¦ç管审è¯è¿ç¨çå éå®æãè¿æ¥è¯¥å ¬å¸ä¸ç管æºæç交æµè¡¨æï¼ä¸ç德西é¦æ²»çCOVID-19ç¸å ³çç³è¯·å审è¯é½å°è¢«å å¿«ã
å¨3æåï¼åå©å¾·ç§å¦åFDA寻æ±è·å¾å¤å¿è¯èµæ ¼ï¼å¼åç德西é¦ä½ä¸ºæ²»çCOVID-19çæ½å¨çæ³ãå¤å¿è¯èµæ ¼æ¯FDAé¼å±å¼åæ²»çç½è§ç çæªæ½ä¹ä¸ï¼ä¸ºå»è¯å ¬å¸å¼åå¨ç çæ³æä¾å¤ç§ä¼æ æ¿çãå ¶ä¸ä¸é¡¹ä¼æ æ¯å¯ä»¥å å»å¨æ°è¯ç³è¯·åé交å¿ç§ç 究计åï¼pediatric study planï¼ãè¿ä¸è¿ç¨ç审è¯æ¶é´å¯è½é¿è¾¾210天ã
åå©å¾·ç§å¦å ¬å¸å¨å£°æä¸è¡¨ç¤ºï¼â åå©å¾·è®¤è¯å°COVID-19大æµè¡ç 带æ¥çç´§æ¥å ¬å ±å«çéæ±ãå ¬å¸æ£å¨ä»¥å°½å¯è½å¿«çé度æ¨è¿ç德西é¦çå¼åãâ
ç德西é¦æ¯åå°å¹¿æ³å ³æ³¨çæç æ¯å¨ç çæ³ï¼å®ç®åå¨6项临åºè¯éªä¸æ¥åæ£éªï¼æ²»çä¸åç±»åçCOVID-19æ£è ãå ¶ä¸ï¼å¨ä¸å½è¿è¡ç两项临åºè¯éªææå¨4æ份è·å¾ç»æã
3æ23æ¥ï¼FDAå®ç½æ°æ®åºæ¾ç¤ºï¼FDAæäºåå©å¾·ç§å¦ï¼Gilead Sciencesï¼å
¬å¸å¼åçå¨ç æç
æ¯çæ³ç德西é¦ï¼remdesivirï¼å¤å¿è¯èµæ ¼ï¼Orphan Drug Designationï¼ï¼å
¶éåºç为å ç¶ç
æ¯ç¾ç
2019ï¼COVID-19ï¼ã
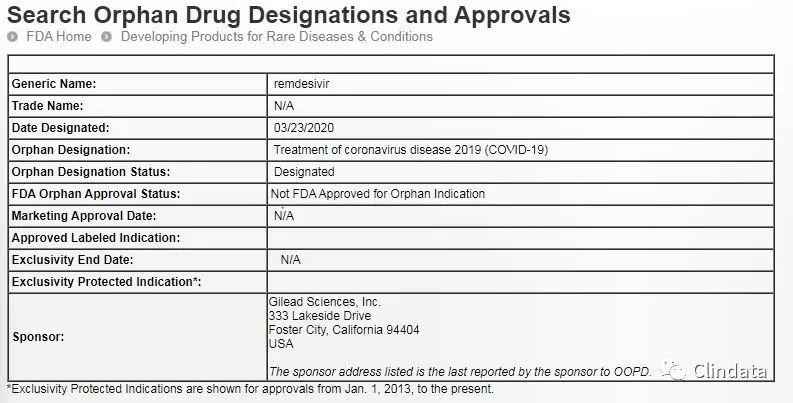
FDAçå¤å¿è¯èµæ ¼è®¤å®é¡¹ç®æ¨å¨ä¿è¿æ²»çç½è§ç¾ç æç¶åµçè¯ç©æçç©å¶åçå¼åãç½è§ç¾ç çå®ä¹æ¯å¨ç¾å½æ£ç 人æ°å°äº20ä¸äººãè·å¾å¤å¿è¯èµæ ¼çå¨ç çæ³è½å¤è·å¾ä¸ç³»åä¿è¿è¯ç©å¼åçä¼æ ï¼å ¶ä¸éè¦çä¸æ¡æ¯å¦æ该çæ³çå¤å¿è¯éåºçè·å¾æ¹åï¼è¯¥è¯ç©å¯ä»¥å¨è¿ä¸éåºçæ¹é¢äº«æ7å¹´å¸åºç¬å æï¼exclusivityï¼ãæå³çå¦æFDAæ¹åç德西é¦ç¨äºæ²»çCOVID-19ï¼7å¹´å å ¶å®å»è¯å ¬å¸å¼åç仿å¶è¯ä¸è½å¨ç¾å½ä¸å¸ãé¤æ¤ä»¥å¤ï¼å¨è¯ç©å¼åè¿ç¨ä¸ï¼ç åå ¬å¸è¿è½å¤è·å¾ä¸åæ ¼ä¸´åºæµè¯ç¸å ³çç¨å¡æµå ï¼ä»¥åé交æ°è¯ç³è¯·æ¶ç¹å®è´¹ç¨çåå çå ¶å®ä¼æ ã
åå©å¾·ç§å¦å ¬å¸å¼åçç德西é¦è¿æ¥æä¸ºå¤§å®¶å ³æ³¨ççç¹è¯ç©ãå®æ¯ä¸æ¬¾æç æ¯çæ³ï¼å ¶ä½ç¨æºå¶ä¸ºæå¶RNAä¾èµæ§RNAèåé ¶çæ´»æ§ï¼ä»èæå¶RNAç æ¯çå¢æ®ãç®åï¼åå©å¾·å ¬å¸æ£å¨è¿å±6项临åºè¯éªï¼æ£éªç德西é¦æ²»çä¸åç±»åçCOVID-19æ£è ççæãå ¶ä¸ï¼å¨ä¸å½è¿è¡ç两项临åºè¯éªææå¨4æè·å¾ç»æã
为äºæ»¡è¶³å¯è½åºç°çæªæ¥éæ±ï¼è¯¥å ¬å¸å·²ç»æ©å¤§ç德西é¦çç产è½åï¼å¹¶ä¸å°ç产两ç§ç德西é¦çé æ¹ï¼æ¶²ä½åå»å¹²ï¼ãæ¨æ¥ï¼è¯¥å ¬å¸è¡¨ç¤ºï¼ç®åæ£å¨å°æä¾ä¸ªäººåæ 使ç¨ï¼compassionate useï¼ç´§æ¥ééè¿æ¸¡å°æ©å±ä½¿ç¨é¡¹ç®ï¼expanded access programsï¼ï¼è¿ç§æ¹æ³å°å ééçæ£è è·å¾ç德西é¦çæºä¼ï¼å¹¶è½å¤æ¶éææåä¸æ£è çæ°æ®ãè¿äºé¡¹ç®ç®åæ£å¨ä¸å ¨çåå½ç管æºæèåå¿«éå¼åã
3æ18æ¥ï¼ä¼ææå¾
çæ´å¹é£é¦-å©æé£é¦æ²»çæ°å 临åºè¯éªç»æï¼å¨ãæ°è±æ ¼å
°å»å¦æå¿ãä¸åå¸ï¼ç»æ让人失æï¼å¨éçæ£è
ä¸ï¼ä¸å¸¸è§æ²»çç¸æ¯ï¼ç 究æªè½è§å¯å°æ´å¹é£é¦-å©æé£é¦æ²»ççæææ§ã
2æåï¼æ¨å¨è¯ä¼°æ´å¹é£é¦-å©æé£é¦æ²»çæ°å ç临åºè¯éªï¼å¨æ¦æ±éé¶æ½å»é¢å¼å±ã
è¯éªå ±çº³å ¥199åæ°å ç æ¯ç¡®è¯æ£è ï¼éæºæ¥å常è§æ²»çç»ï¼å常è§æ²»çå å¹é£é¦-å©æé£é¦æ²»çã临åºæ¹ååç æ¯æ¸ é¤æ åµæ¯ä¸¤ä¸ªå ³é®ææ ï¼ç»ææ¾ç¤ºï¼
临åºçç¶æ¹åï¼ä¸¤ç»æ£è åºç°ä¸´åºç¶åµæ¹åä¸ä½æ¶é´ç¸åï¼å为16天ãæ´å¹é£é¦-å©æé£é¦ç»ä¸å¸¸è§æ²»çç»æ£è çé£é©æ¯ä¸º1.31ï¼95% 置信度ï¼0.95-1.80ï¼ï¼æªè½è¡¨ç°åºæ¾èå·®å¼ã
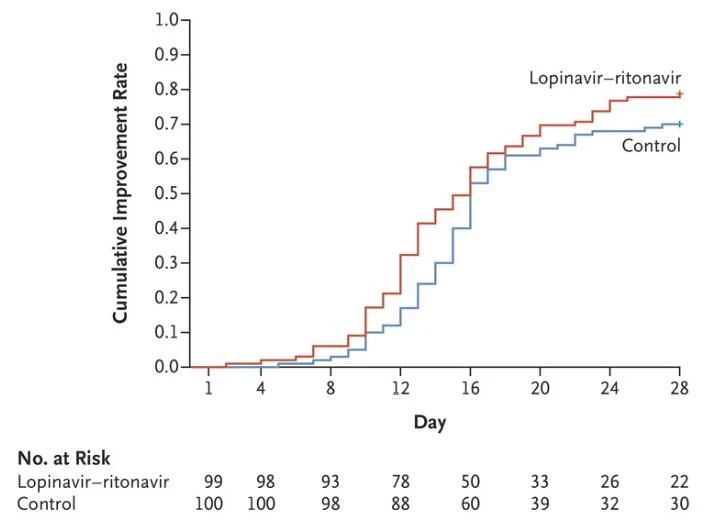
ç
æ¯æ¸
é¤æ
åµï¼ä¸å¸¸è§æ²»çç»ç¸æ¯ï¼æ´å¹é£é¦-å©æé£é¦æ²¡æéä½ç
æ¯RNAè½½éï¼ä¹æ æ³ç¼©çæ£åºç
æ¯RNAçæ¶é´ãè¯éªç»ææ¶ï¼ç¬¬28天ï¼ï¼æ´å¹é£é¦-å©æé£é¦ç»æ40.7%çæ£è
ä»å¯æ£åºæ°å ç
æ¯RNAï¼å常è§æ²»çç»æ å·®å¼ã
æ¤å¤ï¼å¨å®å ¨æ§æ¹é¢ï¼æ´å¹é£é¦-å©æé£é¦ç»æ£è åºç°äº4èµ·èè é严éä¸è¯äºä»¶ï¼å æ¬æ¶å¿ãåååè ¹æ³»ï¼ï¼è常è§æ²»çç»æªåçèè é严éä¸è¯äºä»¶ã
综åç»ææ¯ï¼å常è§æ²»çç¸æ¯ï¼æ´å¹é£é¦-å©æé£é¦æ²»çï¼å¨æ¹å临åºçç¶åæ¸ é¤ç æ¯æ¹é¢å¹¶æ ä¼å¿ï¼è¿æå¯è½å¢å ä¸è¯ååºã
å ³äºæ´å¹é£é¦-å©æé£é¦
æ´å¹é£é¦-å©æé£é¦ï¼åååå åèï¼2000å¹´è·FDAæ¹åä¸å¸ï¼ç¨äºæ²»çè¾æ»ç ã
å¨æ¤åçSARSç«æ åMERSç«æ çååï¼æ´å¹é£é¦-å©æé£é¦é½ä½ä¸ºææ½åçæ²»çè¯ç©ï¼ä½å¨ä¸´åºè¯éªä¸ï¼é½æ²¡æ产çç¡®å¿çææè¯æ®ã
é对COVID-19ï¼æ´å¹é£é¦-å©æé£é¦è¿æ¯æ æãç°å¨ï¼åªè½æå¾
ç德西é¦ä¸´åºæ°æ®äºã
å ³äºäºæ请ç¹å»ï¼ååï¼ç¾å½Remdesivir (ç德西é¦)æ²»çCOVID-19éåºæ§è®¾è®¡æ¥äºï¼äºææå ¥ç»394ä¾éç¨7åç±»æåºåéä½ä¸ºä¸»è¦ç»ç¹
以ä¸æ¯ä¸æ临åºè¯éªæ¹æ¡ï¼
| Arm | Intervention/treatment |
|---|---|
| Experimental: Remdesivir (RDV), 5 Days
Participants will receive continued standard of care therapy together with RDV 200 mg on Day 1 followed by RDV 100 mg on Days 2, 3, 4, and 5. |
Drug: Remdesivir
Administered as an intravenous infusion Other Name: GS-5734™ Drug: Standard of Care Standard of care therapy per local written policies or guidelines |
| Experimental: Remdesivir, 10 Days
Participants will receive continued standard of care therapy together with RDV 200 mg on Day 1 followed by RDV 100 mg on Days 2, 3, 4, 5, 6, 7, 8, 9, and 10. |
Drug: Remdesivir
Administered as an intravenous infusion Other Name: GS-5734™ Drug: Standard of Care Standard of care therapy per local written policies or guidelines |
| Active Comparator: Continued SOC Therapy
Participants will receive continued standard of care therapy. |
Drug: Standard of Care
Standard of care therapy per local written policies or guidelines |
Outcome Measures
Primary Outcome Measures :
Proportion of Participants Discharged by Day 14 [ Time Frame: First dose date or randomization date up to 14 days ]
Secondary Outcome Measures :
Proportion of Participants With Treatment Emergent Adverse Events Leading to Study Drug Discontinuation [ Time Frame: First dose date up to 10 days ]
Eligibility Criteria
| Ages Eligible for Study: | 18 Years and older (Adult, Older Adult) |
| Sexes Eligible for Study: | All |
| Accepts Healthy Volunteers: | No |
Criteria
Key Inclusion Criteria:
Willing and able to provide written informed consent prior to performing study procedures
Severe Acute Respiratory Syndrome Coronavirus (SARS-CoV)-2 infection confirmed by polymerase chain reaction (PCR) test ⤠4 days before randomization
Currently hospitalized with fever defined as temperature ⥠36.6 °C armpit, ⥠37.2 °C oral, or ⥠37.8 °C rectal
Peripheral capillary oxygen saturation (SpO2) > 94% on room air at screening
Radiographic evidence of pulmonary infiltrates
Key Exclusion Criteria:
Participation in any other clinical trial of an experimental treatment for COVID-19
Concurrent treatment with other agents with actual or possible direct acting antiviral activity against SARS-CoV-2 is prohibited < 24 hours prior to study drug dosing
Requiring mechanical ventilation at screening
Alanine Aminotransferase (ALT) or aspartate aminotransferase (AST) > 5 X upper limit of normal (ULN)
Creatinine clearance < 50 mL/min
Note: Other protocol defined Inclusion/Exclusion criteria may apply.
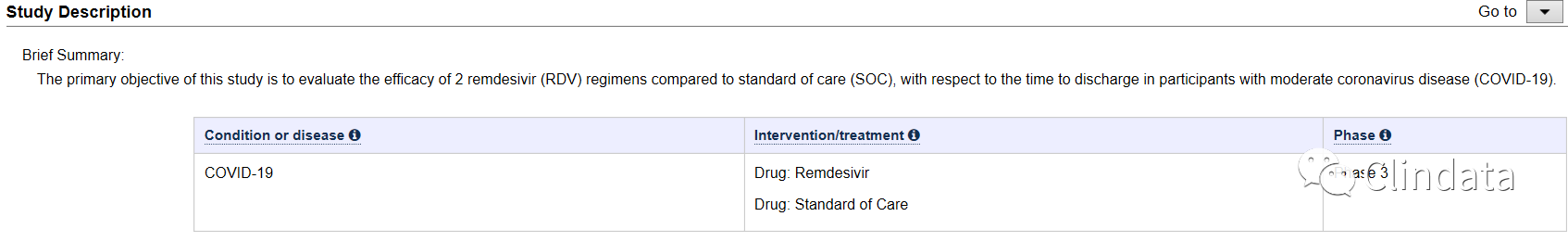
Brief Summary:
The primary objective of this study is to evaluate the efficacy of 2 remdesivir (RDV) regimens compared to standard of care (SOC), with respect to the time to discharge in participants with moderate coronavirus disease (COVID-19).
| Condition or disease | Intervention/treatment | Phase |
|---|---|---|
| COVID-19 | Drug: Remdesivir Drug: Standard of Care | Phase 3 |
Study Design
| Study Type : | Interventional (Clinical Trial) |
| EstimatedEnrollment : | 600 participants |
| Allocation: | Randomized |
| Intervention Model: | Parallel Assignment |
| Masking: | None (Open Label) |
| Primary Purpose: | Treatment |
| Official Title: | A Phase 3 Randomized Study to Evaluate the Safety and Antiviral Activity of Remdesivir (GS-5734™) in Participants With Moderate COVID-19 Compared to Standard of Care Treatment |
| Estimated Study Start Date : | March 2020 |
| Estimated Primary Completion Date : | May 2020 |
| Estimated Study Completion Date : | May 2020 |
Arms and Interventions
| Arm | Intervention/treatment |
|---|---|
| Experimental: Remdesivir (RDV), 5 Days
Participants will receive continued standard of care therapy together with RDV 200 mg on Day 1 followed by RDV 100 mg on Days 2, 3, 4, and 5. |
Drug: Remdesivir
Administered as an intravenous infusion Other Name: GS-5734™ Drug: Standard of Care Standard of care therapy per local written policies or guidelines |
| Experimental: Remdesivir, 10 Days
Participants will receive continued standard of care therapy together with RDV 200 mg on Day 1 followed by RDV 100 mg on Days 2, 3, 4, 5, 6, 7, 8, 9, and 10. |
Drug: Remdesivir
Administered as an intravenous infusion Other Name: GS-5734™ Drug: Standard of Care Standard of care therapy per local written policies or guidelines |
| Active Comparator: Continued SOC Therapy
Participants will receive continued standard of care therapy. |
Drug: Standard of Care
Standard of care therapy per local written policies or guidelines |
Outcome Measures
Primary Outcome Measures :
Proportion of Participants Discharged by Day 14 [ Time Frame: First dose date or randomization date up to 14 days ]
Secondary Outcome Measures :
Proportion of Participants With Treatment Emergent Adverse Events Leading to Study Drug Discontinuation [ Time Frame: First dose date up to 10 days ]
Eligibility Criteria
| Ages Eligible for Study: | 18 Years and older (Adult, Older Adult) |
| Sexes Eligible for Study: | All |
| Accepts Healthy Volunteers: | No |
Criteria
Key Inclusion Criteria:
Willing and able to provide written informed consent prior to performing study procedures
Severe Acute Respiratory Syndrome Coronavirus (SARS-CoV)-2 infection confirmed by polymerase chain reaction (PCR) test ⤠4 days before randomization
Currently hospitalized with fever defined as temperature ⥠36.6 °C armpit, ⥠37.2 °C oral, or ⥠37.8 °C rectal
Peripheral capillary oxygen saturation (SpO2) > 94% on room air at screening
Radiographic evidence of pulmonary infiltrates
Key Exclusion Criteria:
Participation in any other clinical trial of an experimental treatment for COVID-19
Concurrent treatment with other agents with actual or possible direct acting antiviral activity against SARS-CoV-2 is prohibited < 24 hours prior to study drug dosing
Requiring mechanical ventilation at screening
Alanine Aminotransferase (ALT) or aspartate aminotransferase (AST) > 5 X upper limit of normal (ULN)
Creatinine clearance < 50 mL/min
Note: Other protocol defined Inclusion/Exclusion criteria may apply.
å¨çº¿åå

éç¥äº¬ä¸å è¡æ¢ç滴丸

ç¾å©é禾åº

æ®å®åº·å®£èºæ¢å½åå
æé³åå

æé³